Alkynes are organic molecules made of the functional group carbon-carbon triple bonds and are written in the empirical formula of CnH2n−2. They are unsaturated hydrocarbons. Like alkenes have the suffix –ene, alkynes use the ending –yne; this suffix is used when there is only one alkyne in the molecule. In respect to this, can alkynes have more than one triple bond?
Alkenes are hydrocarbons that contain one or more double bonds, while alkynes contain one or more triple bonds.
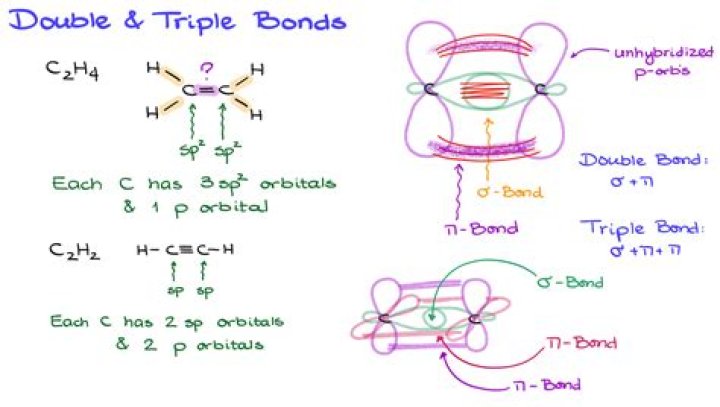
Secondly, what types of bonds are present in an alkyne? Molecules that contain a triple bond between two carbon atoms are known as alkynes. The triple bond is made up of one σ bond and two π bonds.
Besides, can alkenes have triple bonds?
Alkenes can have triple bonds but, they should be between carbon and any other hetero elements and not between carbon and carbon. Otherwise, the compound ceases to be an alkene.
What compound contains a triple bond?
Triple bond, in chemistry, a covalent linkage in which two atoms share three pairs of electrons, as in the nitrogen molecule, N2, or acetylene, C2H2.
Related Question Answers
Which has higher priority double bond or triple bond?
If a molecule contains both a double and a triple bond, the carbon chain is numbered so that the first multiple bond gets a lower number. If both bonds can be assigned the same number, the double bond takes precedence. Do double or triple bonds take priority?
Answer: When a molecule has both a double and triple bond the carbon chain is numbered to give the lowest possible numbers. Meaning double and triple bonds have equal priority. What is the simplest alkyne?
The simplest alkyne—a hydrocarbon with carbon-to-carbon triple bond—has the molecular formula C2H2 and is known by its common name—acetylene (Figure 13.6. Acetylene (ethyne) is the simplest member of the alkyne family. Acetylene is used in oxyacetylene torches for cutting and welding metals. What is the old name of alkenes?
The simplest alkene, ethylene (C2H4) (or "ethene" in the IUPAC nomenclature) is the organic compound produced on the largest scale industrially. Aromatic compounds are often drawn as cyclic alkenes, but their structure and properties are sufficiently distinct that they are not classified as alkenes or olefins. Is Co a triple bond?
Carbon monoxide consists of one carbon atom and one oxygen atom, connected by a triple bond that consists of a net two pi bonds and one sigma bond. Why are there no cyclic alkynes?
In organic chemistry, a cycloalkyne is the cyclic analog of an alkyne. Because of the linear nature of the C–C≡C–C alkyne unit, cycloalkynes can be highly strained and can only exist when the number of carbon atoms in the ring is great enough to provide the flexibility necessary to accommodate this geometry. How many pi bonds are in a triple bond?
two pi bonds
Why are triple bonds linear?
A triple bond forces a linear configuration because of the fact that it is comprised of a sigma and two pi bonds which are rigid, meaning that it cannot rotate. A linear configuration is one where there are no "bends" in the molecular structure of the compound; it keeps going in the same direction. Why are double bonds more reactive than single bonds?
Double bonds are often found in alkenes and ring structures, where the double bond gives more stability due to resonance. They are more reactive than single bonds because they are more electron rich. Why alkene is more reactive than alkanes?
The number of hydrogen atoms in an alkene is double the number of carbon atoms, so they have the general formula. Alkenes are unsaturated, meaning they contain a double bond . This bond is why the alkenes are more reactive than the alkanes . Which is more reactive alkene or alkyne?
1) In acid-base reactions, alkynes are the most reactive followed by alkenes and alkanes. This is due to the stability of conjugate base of alkyne by sp hybridized carbon atom. 2)In electrophilic substitution, alkynes are less reactive than alkenes. Why are triple bonds more reactive?
A triple bond is more reactive than a double bond. That is because it takes energy to form a bond. So, each bond you add between two atoms, increases the potential energy stored in that bond. And, the more potential energy that is stored in a bond, the less stable (more reactive) it is. What are the first 10 alkynes?
Here are the molecular formulas and names of the
first ten carbon straight chain
alkynes.
Introduction.
| Name | Molecular Formula |
| Ethyne | C2H2 |
| Propyne | C3H4 |
| 1-Butyne | C4H6 |
| 1-Pentyne | C5H8 |
What are the first 10 alkenes?
List of Alkenes - Ethene (C2H4)
- Propene (C3H6)
- Butene (C4H8)
- Pentene (C5H10)
- Hexene (C6H12)
- Heptene (C7H14)
- Octene (C8H16)
- Nonene (C9H18)
How can you distinguish between alkyne and alkene?
Alkenes are hydrocarbons that contain one or more double bonds, while alkynes contain one or more triple bonds. The naming conventions for these compounds are similar to those for alkanes. Do alkanes have double bonds?
Alkanes, also called paraffins, are a class of hydrocarbons that are fully saturated with hydrogen. They contain no double or triple bonds in their carbon skeletons and, therefore, have the maximum number of carbon to hydrogen covalent bonds. Why are alkenes very reactive?
Alkenes are relatively stable compounds, but are more reactive than alkanes because of the reactivity of the carbon–carbon π-bond. Because the carbon-carbon π bond is relatively weak, it is quite reactive and can be easily broken and reagents can be added to carbon. What type of bonds are present in alkenes?
An alkene is a hydrocarbon with one or more carbon-carbon double covalent bonds. The simplest alkene is composed of two carbon atoms and is called ethene (shown below). Each carbon is bonded to two hydrogen atoms in addition to the double bond between them. How many bonds are there in alkenes?
Alkenes are hydrocarbons that contain one or more double bonds, while alkynes contain one or more triple bonds. The naming conventions for these compounds are similar to those for alkanes. Is Propyne an alkyne?
Propyne is an alkyne, a terminal acetylenic compound and a gas molecular entity. What type of bonds do alkanes have?
Alkanes have only single bonds, alkenes contain a carbon-carbon double bond, and alkynes contain a carbon-carbon triple bond. Aromatic hydrocarbons make up a group of related substances obtained by chemical breakdown of certain pleasant-smelling plant extracts. How many bonds do alkanes have?
Alkanes, or saturated hydrocarbons, contain only single covalent bonds between carbon atoms. Each of the carbon atoms in an alkane has sp3 hybrid orbitals and is bonded to four other atoms, each of which is either carbon or hydrogen. Why alkyne is called acetylene?
Since the compound is unsaturated with respect to hydrogen atoms, the extra electrons are shared between 2 carbon atoms forming double bonds. Alkynes are also generally known as ACETYLENES from the first compound in the series. Acetylene can be produced from the reaction of solid calcium carbide and water. Which metal is used to convert Dihalides to alkynes?
In general, chlorine or bromine is used with an inert halogenated solvent like chloromethane to create a vicinal dihalide from an alkene. What is a terminal alkyne?
Terminal alkyne: An alkyne in which the carbon-carbon triple bond is at the end of the carbon chain. Are alkynes soluble in water?
Alkynes are nonpolar, unsaturated hydrocarbons with physical properties similar to alkanes and alkenes. Alkynes dissolve in organic solvents, have slight solubility in polar solvents, and are insoluble in water. Compared to alkanes and alkenes, alkynes have slightly higher boiling points. Why are triple bonds strongest?
Triple bonds are stronger than double bonds due to the the presence of two [latex]pi[/latex] bonds rather than one. Each carbon has two sp hybrid orbitals, and one of them overlaps with its corresponding one from the other carbon atom to form an sp-sp sigma bond. Which bond is the strongest?
Two of the strongest forms of chemical bond are the ionic and the covalent bonds. Chemical bonds form between two atoms, each with its own electron environment. Are triple bonds the strongest?
Triple bonds are stronger than the equivalent single bonds or double bonds, with a bond order of three. The most common triple bond, that between two carbon atoms, can be found in alkynes. Other functional groups containing a triple bond are cyanides and isocyanides. Which is the strongest a single double triple bond?
The stronger the bond is, the shorter it will be. The triple bonds are the strongest and hence the shortest. Then comes double bonds which are of intermediate strength between the triple and single bonds. And finally the single bonds are weaker than the other two. Which has a triple bond c2h4 or c3h4?
Answer. C3H4 is the correct option. Because C3H4(Propyne) belongs to the functional group of triple bond,i.e,Alkynes.It has triple bond. Is a triple bond sigma or pi?
In general, single bonds between atoms are always sigma bonds. Double bonds are comprised of one sigma and one pi bond. Triple bonds are comprised of one sigma bond and two pi bonds.