Long chain polypeptides can be formed by linking many amino acids to each other via peptide bonds. The amide bond can only be broken by amide hydrolysis, where the bonds are cleaved with the addition of a water molecule. The peptide bonds of proteins are metastable, and will break spontaneously in a slow process. Keeping this in consideration, how do you break a peptide bond?
A peptide bond can be broken by hydrolysis (the addition of water). In the presence of water they will break down and release 8–16 kilojoule/mol (2–4 kcal/mol) of Gibbs energy. This process is extremely slow, with the half life at 25 °C of between 350 and 600 years per bond.
Also Know, why are peptide bonds stable? The stability of the peptide bond is due to the resonance of amides. With resonance, the nitrogen is able to donate its lone pair of electrons to the carbonyl carbon and push electrons from the carbonyl double bond towards the oxygen, forming the oxygen anion.
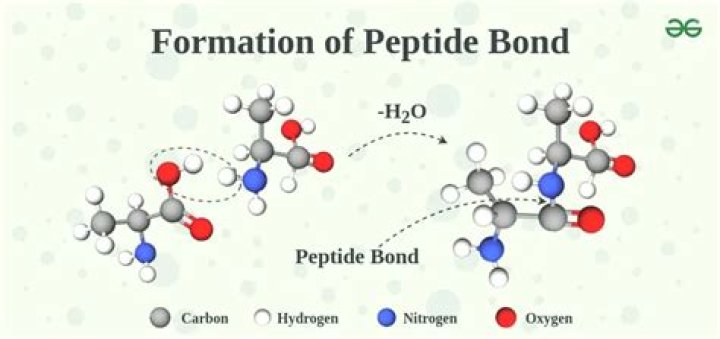
Subsequently, one may also ask, how a peptide bond is formed?
A peptide bond is a chemical bond formed between two molecules when the carboxyl group of one molecule reacts with the amino group of the other molecule, releasing a molecule of water (H2O). This is a dehydration synthesis reaction (also known as a condensation reaction), and usually occurs between amino acids.
Can peptide bonds be broken by heat?
Heat will not alter the order of amino acids in the chain (the primary structure), because it will not effectively break the peptide bonds between them. However, heat can force the bonds between the many polypeptide chains forming the quaternary structure to break.
Related Question Answers
What is an example of a peptide bond?
For instance, a dipeptide is a peptide made up of two amino acids. A tripeptide is a peptide consisting of three amino acids. A peptide bond is sometimes referred to as eupeptide bond since it is the more predominant type. What are the characteristics of a peptide bond?
A peptide bond is a planar, trans and rigid configuration. It also shows a partial double bond character. The coplanarity of the peptide bond denotes the resonance or partial sharing of two pairs of electrons between the amide nitrogen and carboxyl oxygen. What is another name for a peptide bond?
Peptide bond. The resulting CO-NH bond is called a peptide bond, and the resulting molecule is an amide. The four-atom functional group -C(=O)NH- is called an amide group or (in the context of proteins) a peptide group. What are the two types of β sheets?
There are two types of beta sheets, parallel and anti-parallel sheets. Parallel beta sheets are chains of polypeptides, which run in the same direction. Anti-parallel beta sheets are chains of polypeptides which run in opposite directions to each other. What is removed to form a peptide bond between two amino acids?
Peptide bond. From The School of Biomedical Sciences Wiki. Peptide bonds (also known as amide bonds) are the bonds that are found between two monomer units of amino acids in a polypeptide chain. They are created through condensation reactions in which a molecule of water (H2O) is removed. When two amino acids react to form a peptide bond What new functional group is created?
A peptide bond is a chemical bond formed between two molecules when the carboxyl group of one molecule reacts with the amino group of the other molecule, releasing a molecule of water (H2O). This is a dehydration synthesis reaction (also known as a condensation reaction), and usually occurs between amino acids. When an egg is fried what happens to the protein in the egg?
When an egg id fried, what happens to the protein in the egg? Because the heat removes water, the hydrophilic amino acids leave the pan. The amino acids form new proteins. The protein is denatured. Why does a peptide bond resist rotation?
This means that the peptide bond (the C=O. and N-H) all reside in a single plane. Thus, there is no rotation around the bond. Because of the partial double bond between the α carbon and the amine nitrogen, no rotation is possible around that bond. Are peptide bonds strong?
The peptide bond takes on a pseudo-double bond characteristic; rigid, planar, and stronger than a typical C-N single bond. The strength of the peptide bond is largely attributable to the resonance between nitrogen and the carbonyl group. How do you identify a peptide bond?
Amino acids are joined together in proteins by peptide bonds. A peptide bond forms between the carboxyl group of one amino acid (amino acid 1 in the figure below) and the amino group of the adjacent amino acid (amino acid 2). What is the function of peptides?
peptide. A peptide is a short chain of amino acids. Proteins can be digested by enzymes (other proteins) into short peptide fragments. Among cells, peptides can perform biological functions. For example, some peptides act as hormones, which are molecules that when released from cells affect other areas of the body. How many peptide bonds are in a tripeptide?
A tripeptide is a peptide derived from three amino acids joined by two or sometimes three peptide bonds. As for proteins, the function of peptides is determined by the consistuent amino acids and their sequence. The simplest tripeptide is glycylglycylglycine. What happens when two amino acids join together?
Polypeptides and the Peptide bond when joining two amino acids together cells carry out a chemical reaction between the amine group of one amino acid and the carboxylic acid group of the second amino acid. What kind of bond is a peptide bond?
A peptide bond is an amide type of covalent chemical bond linking two consecutive alpha-amino acids from C1 (carbon number one) of one alpha-amino acid and N2 (nitrogen number two) of another along a peptide or protein chain. Is a hydrogen bond?
A hydrogen bond (often informally abbreviated H-bond) is a partial intermolecular bonding interaction between a lone pair on an electron rich donor atom, particularly the second-row elements nitrogen (N), oxygen (O), or fluorine (F), and the antibonding molecular orbital of a bond between hydrogen (H) and a more How do you make peptides?
In organic chemistry, peptide synthesis is the production of peptides, compounds where multiple amino acids are linked via amide bonds, also known as peptide bonds. Peptides are chemically synthesized by the condensation reaction of the carboxyl group of one amino acid to the amino group of another. What happens when a peptide bond is formed?
A peptide bond is a chemical bond formed between two molecules when the carboxyl group of one molecule reacts with the amino group of the other molecule, releasing a molecule of water (H2O). This is a dehydration synthesis reaction (also known as a condensation reaction), and usually occurs between amino acids. What do peptide bonds hold together?
The bond that holds together the two amino acids is a peptide bond, or a covalent chemical bond between two compounds (in this case, two amino acids). It occurs when the carboxylic group of one molecule reacts with the amino group of the other molecule, linking the two molecules and releasing a water molecule. Do peptide bonds carry a charge?
A peptide bond is a linkage between the building blocks of proteins called amino acids (shorter strings of linked amino acids are known as peptides). The O atom of the amide has a partial negative charge and is a good hydrogen bond acceptor, while the NH is partially positive and a good hydrogen bond donor. Why are peptide bonds important for the secondary structure of proteins?
Basically, the way they have limited rotation, the way they hydrogen-bond to each other in a specific pattern those factors provide the "leverage" over what kind of secondary structure the protein takes up. Its ~40% double bond character makes it rigid, limiting the Cα−N bond rotation. Do all proteins have disulfide bonds?
Disulfide Bonds in Proteins Disulfide bonds in protein membranes are found in both bacteria and eukaryotes. Extracellular proteins often have several disulfide bonds, whereas intracellular proteins usually lack them. In proteins, these bonds form between the thiol groups of two cysteine amino acids. What does Ramachandran plot tell you?
The Ramachandran plot is a plot of the torsional angles - phi (φ)and psi (ψ) - of the residues (amino acids) contained in a peptide. By making a Ramachandran plot, protein structural scientists can determine which torsional angles are permitted and can obtain insight into the structure of peptides. How do you bond amino acids?
Each amino acid is attached to another amino acid by a covalent bond, known as a peptide bond. When two amino acids are covalently attached by a peptide bond, the carboxyl group of one amino acid and the amino group of the incoming amino acid combine and release a molecule of water. When two amino acids are linked together a peptide bond is formed between the?
Section 3.2Primary Structure: Amino Acids Are Linked by Peptide Bonds to Form Polypeptide Chains. Proteins are linear polymers formed by linking the α-carboxyl group of one amino acid to the α-amino group of another amino acid with a peptide bond (also called an amide bond). Does Heating whey protein destroy it?
So to sum it up, yes, cooking with protein powder is completely fine. The heat will denature the protein content slightly, but this has no negative impact on the overall functionality of the protein once it enters your body. Which level of protein structure will not be broken by too much heat?
Heat will not alter the order of amino acids in the chain (the primary structure), because it will not effectively break the peptide bonds between them. However, heat can force the bonds between the many polypeptide chains forming the quaternary structure to break. What are the 4 levels of protein structure?
Four levels of protein structure. The four levels of protein structure are primary, secondary, tertiary, and quaternary. It is helpful to understand the nature and function of each level of protein structure in order to fully understand how a protein works. What molecule would your body have to add to the dipeptide in order to break it down?
Two amino acids can be joined together by dehydration condensation to form a dipeptide. In the laboratory, we can break, or hydrolyze, peptide bonds most effectively by a combination of heat and acid. Why are the terms protein and polypeptide not interchangeable?
The difference is between the structure and the function. While a protein is made of polypeptide bonds between amino acids, it has been structured in a way that causes it to have a function. The polypeptide is when the protein has not fully formed towards it's active state as a protein. At what temperature do proteins denature?
105.8°F
Is denaturation pH reversible?
Reversibility and irreversibility In many cases, denaturation is reversible (the proteins can regain their native state when the denaturing influence is removed). This process can be called renaturation. What is the name of the bond that is created when two amino acids join?
Peptide Bonds