Also question is, what is calorimeter measured in?
Where q is the amount of heat according to the change in temperature measured in joules and Cv is the heat capacity of the calorimeter which is a value associated with each individual apparatus in units of energy per temperature (joules/kelvin).
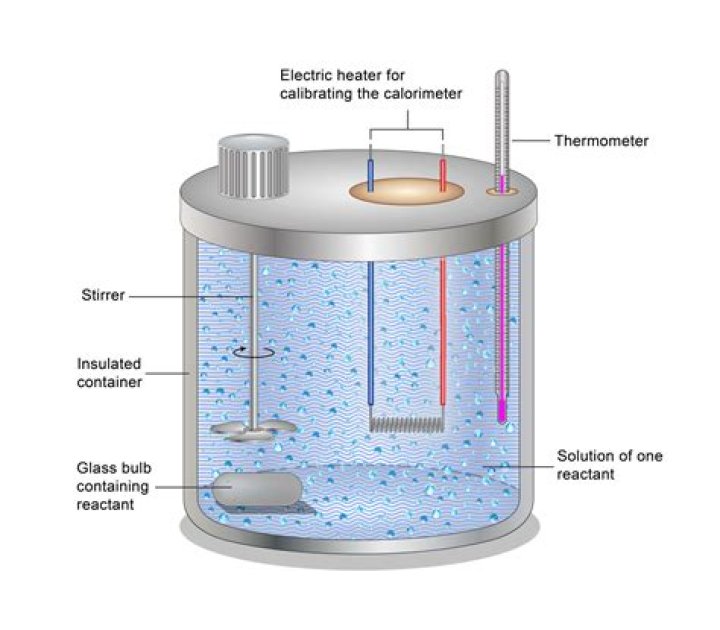
Also Know, does a calorimeter measure energy? The calorimeter traps all the heat from a chemical reaction, we measure the effect of that heat on the temperature of water in the calorimeter, and we can then calculate the heat energy released by the reaction. The calorimeter is an insulated container, in which we place a measured mass of water.
Likewise, does a calorimeter measure heat capacity?
The heat capacity of the calorimeter is the quantity of heat absorbed by the calorimeter for each 1°C rise in temperature. The heat capacity of the calorimeter must be determined experimentally. The easiest process is to study the mixing of warm and cold water.
What is heat measured in?
Two classes of instruments are employed for the measurement of heat, namely, thermometers and pyrometers. Thermometers are only employed to measure comparatively low temperatures, and we shall confine our remarks entirely to this class in the present article.
Related Question Answers
Who invented calorimeter?
Antoine LavoisierWhy is bomb calorimeter called so?
Bomb calorimeter consists of a strong steel vessel ( called bomb)which can stand high pressure when the substance is burnt in it. Hence, it is called bomb calorimeter.How do I calculate specific heat?
The heat capacity and the specific heat are related by C=cm or c=C/m. The mass m, specific heat c, change in temperature ΔT, and heat added (or subtracted) Q are related by the equation: Q=mcΔT. Values of specific heat are dependent on the properties and phase of a given substance.What is the principle of calorimeter?
Ans. The principle of calorimetry states that heat loss from one object is equivalent to heat gain by another object.Why is a calorimeter more accurate?
More reliable results can be obtained by repeating the experiment many times. The biggest source of error in calorimetry is usually unwanted heat loss to the surroundings. This can be reduced by insulating the sides of the calorimeter and adding a lid.What are the types of calorimeter?
The most common types of calorimeters are differential scanning calorimeters, titration calorimeters, isothermal micro calorimeters, and accelerated rate calorimeters. A normal calorimeter usually consists of a thermometer.How does a calorimeter measure heat?
A calorimeter measures the change in heat. Simple calorimeters are made with a metal container of water, positioned above a combustion chamber. The calorimeter measures the mass of the liquid along with the temperature change, to determine the amount of energy change.Which unit expresses heat capacity?
the heat capacity of a defined system is the amount of heat (usually expressed in calories, kilocalories, or joules) needed to raise the system's temperature by one degree (usually expressed in Celsius or Kelvin). It is expressed in units of thermal energy per degree temperature.Can heat capacity be negative?
Most physical systems exhibit a positive heat capacity. However, even though it can seem paradoxical at first, there are some systems for which the heat capacity is negative. A negative heat capacity can result in a negative temperature.What does the heat capacity of an object measure?
Heat capacity is a measure of the amount of heat energy required to change the temperature of a pure substance by a given amount.How is heat flow measured?
Heat flow is calculated using the rock thermal conductivity multiplied by the temperature gradient. The standard units are mW/m2 = milli Watts per meter squared. Thus, think of a flat plane 1 meter by 1 meter and how much energy is transferred through that plane is the amount of heat flow.What is the difference between specific heat capacity and heat capacity?
Heat capacity is the ratio of the amount of heat energy transferred to an object to the resulting increase in its temperature. Specific heat capacity is a measure of the amount of heat necessary to raise the temperature of one gram of a pure substance by one degree K.What makes a good calorimeter?
The calorimeter is a small container made of a metal, such as gold or copper, with good thermal conductivity. It has a well for a temperature sensor, which always has to be at exactly the same temperature as the calorimeter and its contents.What does a calorimeter directly measure?
A calorimeter directly measures temperature change and this information, along with the substance's mass and specific heat, is used to calculate the amount of heat.Why does a calorimeter have two cups?
Commercial solution calorimeters are also available. Relatively inexpensive calorimeters often consist of two thin-walled cups that are nested in a way that minimizes thermal contact during use, along with an insulated cover, handheld stirrer, and simple thermometer.How does a calorimeter work biology?
A calorimeter is a machine used in the process of calorimetry . Small samples of material are placed in the machine which burns them. The energy given off when the material burns is measured. The change in temperature of the water is a direct measure of the energy held within the sample.What are 3 types of heat?
The three types of heat transfer- Convection.
- Conduction.
- Radiation.