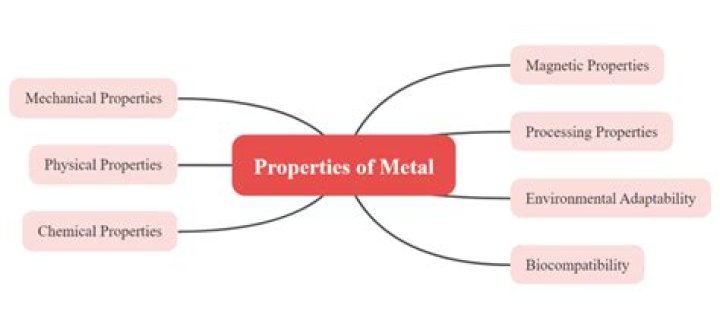
Metals are lustrous, malleable, ductile, good conductors of heat and electricity. Other properties include: State: Metals are solids at room temperature with the exception of mercury, which is liquid at room temperature (Gallium is liquid on hot days). Ductility: Metals can be drawn into wires.
Also asked, what are the 5 properties of metal?
Metal Physical Properties:
- Lustrous (shiny)
- Good conductors of heat and electricity.
- High melting point.
- High density (heavy for their size)
- Malleable (can be hammered)
- Ductile (can be drawn into wires)
- Usually solid at room temperature (an exception is mercury)
- Opaque as a thin sheet (can't see through metals)
One may also ask, which of the following elements is a metal? Most elements can be considered metals. They are grouped together in the middle to the left-hand side of the periodic table. The metals consist of the alkali metals, alkaline earths, transition metals, lanthanides, and actinides.
List of Metals.
| NUMBER | SYMBOL | ELEMENT |
|---|---|---|
| 4 | Be | Beryllium |
| 11 | Na | Sodium |
| 12 | Mg | Magnesium |
| 13 | Al | Aluminum |
Consequently, what are the 10 properties of metals?
Properties of Metals
- Shiny (lustrous) in nature.
- Metal is a good conductor of electricity and heat.
- Density and melting point is high.
- Mouldable (Malleable)
- Ductile.
- At room temperature, it is in solid form except for mercury.
- Opaque.
What is metal or non metal?
nonmetal. The elements can be classified as metals, nonmetals, or metalloids. Metals are good conductors of heat and electricity, and are malleable (they can be hammered into sheets) and ductile (they can be drawn into wire).
Related Question Answers
What are the 6 properties of metal?
Metal Physical Properties:- Lustrous (shiny)
- Good conductors of heat and electricity.
- High melting point.
- High density (heavy for their size)
- Malleable (can be hammered)
- Ductile (can be drawn into wires)
- Usually solid at room temperature (an exception is mercury)
- Opaque as a thin sheet (can't see through metals)
What are the types of metal?
Types of Metals- Aluminum. Aluminum has a dull silver finish, making it similar in appearance to stainless steel.
- Stainless Steel. An alloy of carbon, iron, chromium, and other metals, stainless steel is found in furniture and fixtures throughout the home – from accent tables to kitchen sinks.
- Brass.
- Copper.
- Wrought Iron.
What are the 7 properties of metal?
Metal Physical Properties:- Lustrous (shiny)
- Good conductors of heat and electricity.
- High melting point.
- High density (heavy for their size)
- Malleable (can be hammered)
- Ductile (can be drawn into wires)
- Usually solid at room temperature (an exception is mercury)
- Opaque as a thin sheet (can't see through metals)
What are three properties of metal?
Three properties of metals are:- Luster: Metals are shiny when cut, scratched, or polished.
- Malleability: Metals are strong but malleable, which means that they can be easily bent or shaped.
- Conductivity: Metals are excellent conductors of electricity and heat.
What are the 5 metallic properties and explanation?
(ii) Ductility , the ability to be drawn out into a wire. (iii) Thermal conductivity , the ability to conduct heat. (iv) Electrical conductivity , the ability to conduct electricity. (v) Chemical reactivity , metals tend to be good reducing agents.What properties do all metals have in common?
The following are the common properties shared by the metals : Metals are :- Good electrical conductors and heat conductors.
- Malleable - can be beaten into thin sheets.
- Ductile - can be stretched into wire.
- Possess metallic luster.
- Opaque as thin sheet.
- Solid at room temperature (except Hg).
- They possess good strength.
Is silica a metal?
It's usually found linked up with a pair of oxygen molecules as silicon dioxide, otherwise known as silica. Quartz, an abundant ingredient in sand, is made up of non-crystallized silica. Silicon is neither metal nor non-metal; it's a metalloid, an element that falls somewhere between the two.Which metal is the best conductor of electricity?
Silver also has the highest thermal conductivity of any element and the highest light reflectance. Although it is the best conductor, copper and gold are used more often in electrical applications because copper is less expensive and gold has a much higher corrosion resistance.What is a property of most metals?
Most elements are metals. All metals except for mercury are solids at room temperature. Many metals are shiny, ductile, and malleable. Most are also good conductors of heat.What are the major difference between metals and non metals?
While metals are good conductors of electricity and heat, non-metals are poor conductors. Unlike non-metals, metals are ductile, which means they can be stretched into wires. Another difference between metals and non-metals, is that the former has a metallic luster, while the other does not.What is metal and non metal Class 8?
Materials: Metals and Non-Metals Class 8 Science NCERT Textbook Questions| Properties | Metals | Non-metals |
|---|---|---|
| 3. Malleability | malleable | non-malleable |
| 4. Ductility | ductile | non-ductile |
| 5. Heat Conduction | good conductors | bad conductors |
| 6. Conduction of Electricity | good conductors | bad conductors/insulators |